Aquí hay algunas preguntas y respuestas frecuentes que tocan importantes actualizaciones relacionadas con COVID-19.
Aquí hay algunas preguntas y respuestas frecuentes que tocan importantes actualizaciones relacionadas con COVID-19.

Después de enviar una solicitud y recibir un presupuesto de nuestro Equipo de Apoyo al Paciente, los pacientes suelen tener muchas preguntas sobre el precio que reciben. En este artículo, puede encontrar un desglose del coste total de un medicamento al que le estamos ayudando a acceder, además de recursos sobre cómo puede conseguir que se le reembolse.

Para ayudarle a agilizar el proceso de solicitud con nosotros, hemos elaborado una guía de prescripción rápida.

5 preguntas comunes sobre el suplemento que puede ser útil para la ELA, el Parkinson y el Alzheimer.

Un nuevo análisis del aducanumab de Biogen muestra una reducción en el declive clínico de la enfermedad de Alzheimer.
Como pioneros de un tipo completamente nuevo de servicio mundial, entendemos que la confianza, la seguridad y la credibilidad son primordiales, especialmente cuando se trata de pagar por medicamentos que son esenciales y a menudo la única opción de tratamiento disponible. Hay un número cada vez mayor de sitios web de estafas y nosotros mismos hemos sido objeto de una de esas estafas. En este artículo le daremos consejos sobre cómo detectar a estos farsantes.

Soy afortunado. Han pasado 4 años desde los primeros síntomas del MND, sólo mi voz se ha visto afectada.

Dos años y medio después de su diagnóstico, David encontró un medicamento con una solicitud de MND fuera de la etiqueta.

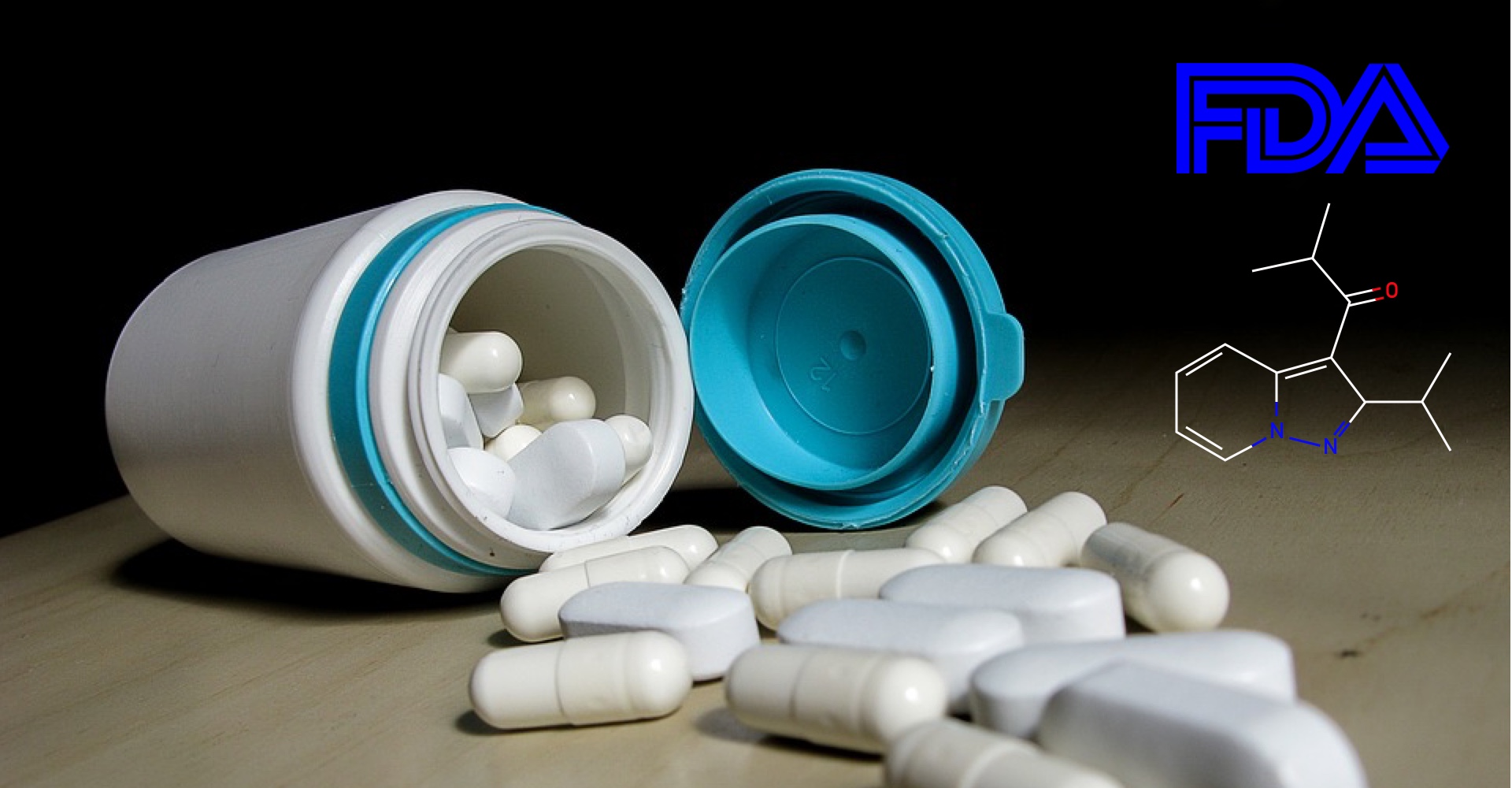
La terapia contra el Parkinson de Kyowa Hakko Kirin, Nourianz (istradefylline), obtiene la aprobación de la FDA como complemento de levodopa/carbidopa.

Radicut/Radicava (edaravone), que ha demostrado ralentizar la progresión de la ELA, está ahora aprobada para pacientes en China.

Orkambi y Symkevi han sido rechazados por el Consorcio de Medicamentos de Escocia (SMC) debido a su conclusión de que los costes superan los beneficios.

Vitrakvi (larotrectinib) recomendado por el CHMP para su aprobación en la UE.

 Después de 40 años con colangitis biliar primaria, la madre de Fiona sigue viva gracias a la dedicación de su hija y a un transplante de hígado.
Después de 40 años con colangitis biliar primaria, la madre de Fiona sigue viva gracias a la dedicación de su hija y a un transplante de hígado.
Después de 3 años de vivir con ELA, Marc encontró una nueva medicina al otro lado del mundo.
Con lágrimas, risas y esperanza, Mark y su familia encuentran su propio camino para hacer frente a la ELA.

Diagnosticado con ELA hace sólo 6 meses, Peter fue contra la corriente para encontrar más opciones para tratar su enfermedad.

MN-166 (ibudilast) formará parte del ensayo clínico de fase III en sujetos con esclerosis múltiple progresiva secundaria sin recaídas.

Nueva medicina para el cáncer de piel ahora aprobada en la UE para adultos con carcinoma de células escamosas cutáneas metastásico o localmente avanzado.

Seguimos caminando y hablando después de 4 años con ELA: compartimos la experiencia de Bakr de vivir con una enfermedad rara en los Emiratos Árabes Unidos para avanzar en su objetivo de crear conciencia y conectar con otros pacientes con ELA.

El ensayo clínico de fase 2 muestra que ibudilast está relacionado con una ralentización del 48% en la progresión de la atrofia cerebral en comparación con el placebo en pacientes con EM progresiva.
Vyndaqel (tafamidis) está ahora aprobado en los EE.UU. después de que un estudio demostrara un aumento de la tasa de supervivencia y una reducción del tiempo de hospitalización por problemas relacionados con el corazón.

La aprobación de la siguiente fase de los ensayos clínicos pone a los pacientes de ELA un paso más cerca de acceder a un nuevo tratamiento que se ha demostrado que retrasa la progresión de la enfermedad.
Hoja de datos descargable que responde a la pregunta más común de The SocialMedwork hecha por los proveedores de salud.

Basándose en los prometedores resultados de los ensayos clínicos de fase III, el Ministerio de Sanidad de Canadá ha decidido ampliar el uso de Kalydeco (ivacaftor) para incluir a los niños de 1 a 2 años.

En reconocimiento al Día de las Enfermedades Raras, nuestra colega, Sera, comparte su historia de la lucha de su padre contra la Atrofia Multisistémica (MSA).

Un ensayo clínico reciente indica que Symkevi/Symdeko (tezacaftor/ivacaftor) puede aflojar de forma segura y eficaz la mucosidad espesa y pegajosa en niños de 6 a 11 años.

El nuevo medicamento para el diagnóstico del cáncer, Vitrakvi (larotrectinib), obtiene la aprobación acelerada de la Administración de Alimentos y Medicamentos de los Estados Unidos (FDA).

La evaluación del CHMP respalda la aprobación de la EMA del medicamento contra el cáncer de pulmón no microcítico (CPNM), Vizimpro (dacomitinib).

El ensayo de fase III muestra que Copiktra (duvelisib) puede ser una opción para los pacientes con leucemia linfocítica crónica en recaída o refractaria y linfoma linfocítico pequeño.
